Inpatient
Colonoscopy
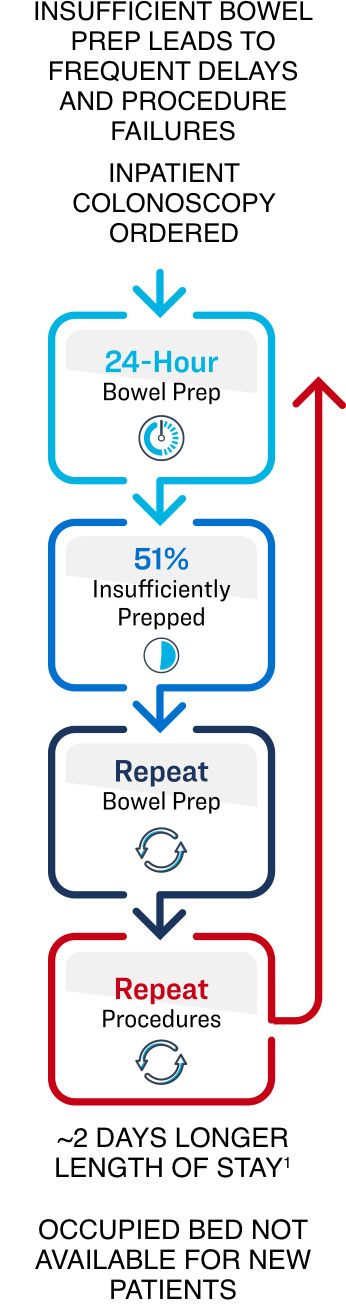
Inpatient colonoscopies are a common occurrence in a hospital setting with nearly 4 million annual procedures worldwide, and are typically preformed to diagnose lower GI bleeds, severe anemia and unknown abdominal pain.1 However, in an emergent situation, the pre-procedural bowel preparation can represent significant challenges where up to 55% of patients can present with an insufficiently prepped colon, leading to longer procedure times, the need for repeat procedures, extended hospital stays and delayed diagnoses, all of which significantly increase costs for hospitals and interfere with quality care for patients.2
Bowel prep challenges represent significant burden





Potential to reduce direct costs for inpatient colonoscopy
We believe the Pure-Vu® System has the ability to overcome these clinical challenges by reliably and predictably moving patients through the hospital system to a successful examination. Since most of inpatient colonoscopies are currently covered under the bundled payment of a Diagnosis-Related Group (DRG) reimbursement, we believe the Pure-Vu® System has the potential to offer a new standard of care which may enhance hospital efficiency by improving quality of care, reducing the length of stay and direct costs, as well as accelerating bed turn-over for new patients. The Pure-Vu® System does not currently have a unique reimbursement code with any private or governmental third-party payors in any country.


The Pure-Vu® System has the potential to:
- Expedite diagnosis and treatment
- Improve the quality of the exam
- Shorten hospital length of stay
- Avoid costs due to prep inefficiencies
Following the recent FDA clearance of the second generation Pure-Vu® System (“Pure-Vu® GEN2”), Motus GI plans to initiate its commercial launch in the U.S. with an initial focus on inpatient colonoscopy, with plans to expand into the high medical need outpatient market. The commercial launch of Pure-Vu® in international markets will be explored in the future.
1. HRA Healthcare Research & Analytics - Market Research, May 2015
2. Yadlapati, R., Johnston, E.R., Gregory, D.L. et al. Predictors of Inadequate Inpatient Colonoscopy Preparation and Its Association with Hospital Length of Stay and Costs. Dig Dis Sci 60, 3482–3490 (2015). https://doi.org/10.1007/s10620-015-3761-2
